Writing New Recipes for High-Performance Materials
Elizabeth Witherspoon
Duke materials scientists and engineers deploy AI and other digital tools to accelerate development of new materials to harvest energy, heal the body and more
New high-performance materials are as vital to meeting complex needs in energy, national security, health care and communications as silicon is to the information age.
Atoms and molecules of natural and synthetic elements are the building blocks of new metal alloys that withstand the temperatures of space travel or improve circuits in electronic devices. Newly discovered polymer nanocomposites have enabled new capacitors and electrodes for the energy grid and flexible electronics. Engineered hydrogels and other biomaterials can help repair human bone or deliver pain medication inside surgical wounds.
Materials scientists and engineers in Duke University’s Thomas Lord Department of Mechanical Engineering & Materials Science (MEMS) are leading research to design and discover these novel materials for our future.
Collaborating across disciplines, they are leveraging the powers of supercomputing, artificial intelligence (AI) and machine learning to cut years off the materials discovery process. They are building open-source platforms to share these data with academic institutions and industry—bringing us closer to solutions for some of society’s toughest problems.
Many have likened the enterprise to developing new recipes for the materials of tomorrow. Below, take a look at what’s cooking in the labs—and minds—of some of Duke MEMS faculty members:

Stefano Curtarolo
Accelerating Discovery of Metal Alloys by Standardizing Data
Professor of Mechanical Engineering & Materials Science
Director, Center for Autonomous Materials Design
Leader, Duke Automatic-FLOW for Materials Database (AFLOW)
Stefano Curtarolo was delighted when the U.S. government launched its Materials Genome Initiative to speed the discovery and dissemination of new materials in 2011, as it increased attention and funding for the work that he had begun almost a decade earlier. Once toiling among only a handful of others worldwide to find new recipes for metal alloys and build a standardized database for sharing information about various material properties, he now leads a consortium of 16 institutions across the globe who continually update this open source that helps researchers build practical new materials.
The Duke Automatic-FLOW for Materials Database (AFLOW) now contains information on 3.3 million material compounds with over half a billion calculated material properties, including a comprehensive update and addition of a database of crystalline lattice structures.
Curtarolo’s group mines these data to run high-throughput computer simulations that have produced new metal alloys, as well as the recipes for two new magnetic materials.
“The ability to rapidly predict the existence of these magnetic materials is a major coup and will be invaluable to materials scientists moving forward,” said Curtarolo.
Most recently, his group identified a potentially promising alloy with a very high melting point that could enable it to substitute for the expensive rare earth metal rhenium in high-temperature applications like rocket engine nozzles.
Curtarolo also will use the AFLOW database, AI and machine learning in his latest project–leading a $7.5-million, five-year Multidisciplinary University Research Initiative (MURI) award from the Office of Naval Research with collaborators from four other universities–to identify super/ultra-hard disorded ceramics with a wide array of potential applications.

Cate Brinson
Finding Hard Answers to Soft Matter Questions
Donald M. Alstadt Chair, Thomas Lord Department of Mechanical Engineering & Materials Science
Sharon C. and Harold L. Yoh, III Professor
Director, AI for Understanding and Designing Materials (aiM)
Cate Brinson’s research combines computational models, data science and physical experiments to lay the groundwork for developing new materials for applications ranging from a more stable power grid to biomedical devices and lightweight structures. An internationally recognized expert in soft matter who joined Duke in 2017, she leads a $5-million National Science Foundation-funded national consortium focused on building a data-driven framework specifically for the discovery of new polymer nanocomposites and structural metamaterials.
“If you have significant portions of the relevant data fully accessible, then you can create a design loop where you start to design materials based on the ideal properties for a given application,” said Brinson. NanoMine is a platform to make this data readily available to other researchers that otherwise would take years to tease out manually from traditional journal resources.
NanoMine is a platform to make this data readily available to other researchers that otherwise would take years to tease out manually from traditional journal resources.
Think of this data framework as analogous to the step before chefs can author their recipes. It will be the reference guide for the recipe writers that explains how different ingredients behave in various combinations and under various conditions. Listen to Brinson explain it in Duke Engineering’s Rate of Change podcast, “In the Kitchen with NanoMine.”
Using this platform along with machine learning tools, her team has demonstrated the power of data and design to find the optimal composition and microstructure of dielectrical (insulating) nanocomposites, which eliminates the traditional tradeoff between a material’s electrical insulating capacity and its ability to prevent current from breaking through, enabling new paradigms in capacitors for energy applications.
Brinson also leads a new $3-million NSF-funded graduate training program, called “AI for Understanding and Designing Materials (aiM).” This program integrates training for PhD students in both materials and computer science, teaching them how to collaborate to advance the frontiers of research and training in the new convergent field leveraging the power of AI.

Volker Blum
Unlocking the Secrets of New Energy Materials
Associate Professor of Mechanical Engineering & Materials Science
Leader, Ab Initio Materials Simulations Group at Duke
Coordinator and Lead Developer, FHI-aims computer platform
Volker Blum’s group uses computational models incorporating the fundamental laws of quantum mechanics to help develop new semiconductor materials for energy-related applications, such as photovoltaic materials or low-cost manufacturable organic-inorganic semiconductors. He works with a range of collaborators at Duke, elsewhere in North Carolina’s Research Triangle region and across the globe.
The U.S. Department of Energy tapped Blum, along with Duke colleagues David Mitzi and Adrienne Stiff-Roberts, and 13 other researchers nationally to better understand and control the emergent phenomena of spin, charge, and light/matter interactions in hybrid organic-inorganic semiconductors to potentially unlock new energy technologies.
In a recent development the group helped demonstrate hybrid semiconductors in which a targeted chiral molecule–a structure that exists in two different, mirror-symmetric versions, in the same way as human hands are mirror-symmetric but not identical–transfers its “handedness” from the organic molecule to the inorganic part of the structure. They showed that this “chirality transfer” enables control over the spin-related character of the semiconductor electronic bands, which is an important step toward using these types of materials in electrical devices that work based on the electrons’ spin.
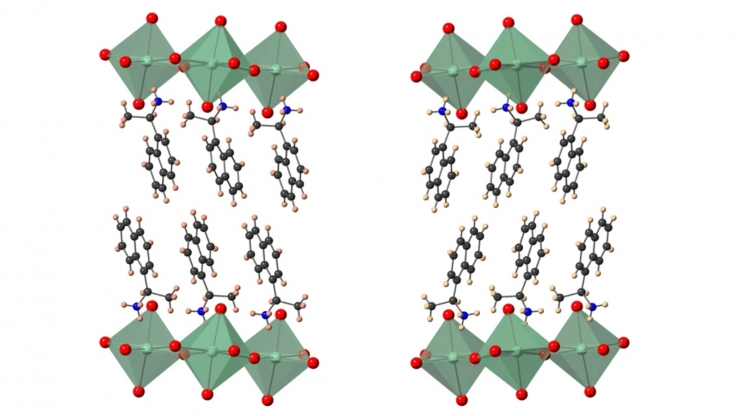 “The materials used for the first hybrid perovskite solar cells were invented in the 1970s by a German laboratory that just wanted to study their properties to see what they could do,” said Blum, who led the computational modeling side of the research. “It wasn’t until 40 years later that someone got the idea to use them for a solar cell. In many ways, that’s what we’re doing here with chiral-induced spin selectivity.”
“The materials used for the first hybrid perovskite solar cells were invented in the 1970s by a German laboratory that just wanted to study their properties to see what they could do,” said Blum, who led the computational modeling side of the research. “It wasn’t until 40 years later that someone got the idea to use them for a solar cell. In many ways, that’s what we’re doing here with chiral-induced spin selectivity.”
In the NSF-funded HybriD3 consortium, Blum and five other principal investigators at Duke, University of North Carolina and North Carolina State University also focus on hybrid organic-inorganic perovskites in a broader materials space exploration. Part of this work includes the creation of an open, curated materials property database, called HybriD3, intended to aid the efforts of other researchers, nationally and internationally, in navigating this promising class of semiconductor materials.
“The FHI-aims program provides a mathematical guide for materials research.”
Blum is also the lead developer and one of the coordinators of the community developing the FHI-aims computer program, which computational materials researchers worldwide use to predict properties of condensed matter (molecules, clusters, solids, liquids, etc.) based on a description of their electronic structure.
“If you want to predict a material’s properties, you need to know what its structure is at given conditions and how it responds to external stimuli like pressure, temperature, light or an electrical field. And to predict all these behaviors, you need to know how to calculate the energy of the materials’ atoms in a particular arrangement, and how this energy changes based on external stimuli,” said Blum. “That is what the FHI-aims program provides–it’s a mathematical guide for materials research.”

Gaurav Arya
Modeling Soft Materials with Hard Calculations
Associate Professor of Mechanical Engineering & Materials Science
Gaurav Arya’s group uses computational methods to provide fundamental, molecular-level understanding of a diverse range of biological and soft material systems. His collaborations reach across the U.S. to address questions critical to materials discovery. He provides materials modeling leadership for an $18-million NSF center developing nanoscale building blocks for self-assembling mesomaterials with emergent properties and on additional $2-million and $1.75-million NSF grants on developing dynamic DNA-based devices for nanoscale sensing and genome engineering applications.
The group recently developed a simplified method for calculating the forces that cause nanoparticles to self-assemble into larger structures. This method enables previously impossible predictions about how nanoparticles with a wide variety of shapes will interact with one another, opening up opportunities for rationally designing such particles for applications in solar energy harvesting and in catalyzing new types of reactions.
“Accurately calculating the forces that pull these particles together at very close range is extremely computationally expensive,” said Arya. “We have now demonstrated an approach that speeds those calculations up by millions of times while only losing a small amount of accuracy.”
“We have now demonstrated an approach that speeds up calculations by millions of times while only losing a small amount of accuracy.”
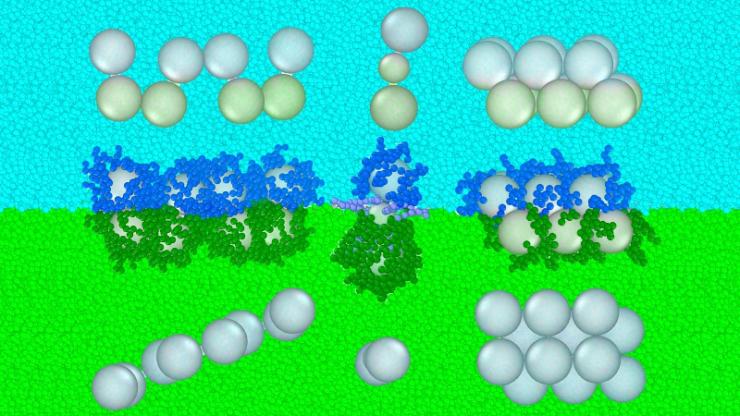 Borrowing another cooking analogy for materials research, Arya also proposed a method for designing nanoparticles that takes advantage of forces in the layers formed by liquids, like those of oil and vinegar in an unshaken bottle of vinaigrette. His idea is to precisely control these competing forces—clustering vs. repelling—to help scientists create novel arrangements in assembly of materials, enabling new combinations with new properties and applications.
Borrowing another cooking analogy for materials research, Arya also proposed a method for designing nanoparticles that takes advantage of forces in the layers formed by liquids, like those of oil and vinegar in an unshaken bottle of vinaigrette. His idea is to precisely control these competing forces—clustering vs. repelling—to help scientists create novel arrangements in assembly of materials, enabling new combinations with new properties and applications.
“There are so many things to do next. For one, we’d like to explore the full repertoire of possible structures and phases researchers could make using this concept. We are also working closely with experimentalists to test the full capabilities of this approach,” said Arya.
Learn more about Duke MEMS Computation & Artificial Intelligence Research
